Research I 6 August 2025
EuroACT Phase 3: Advancing from data to evidence
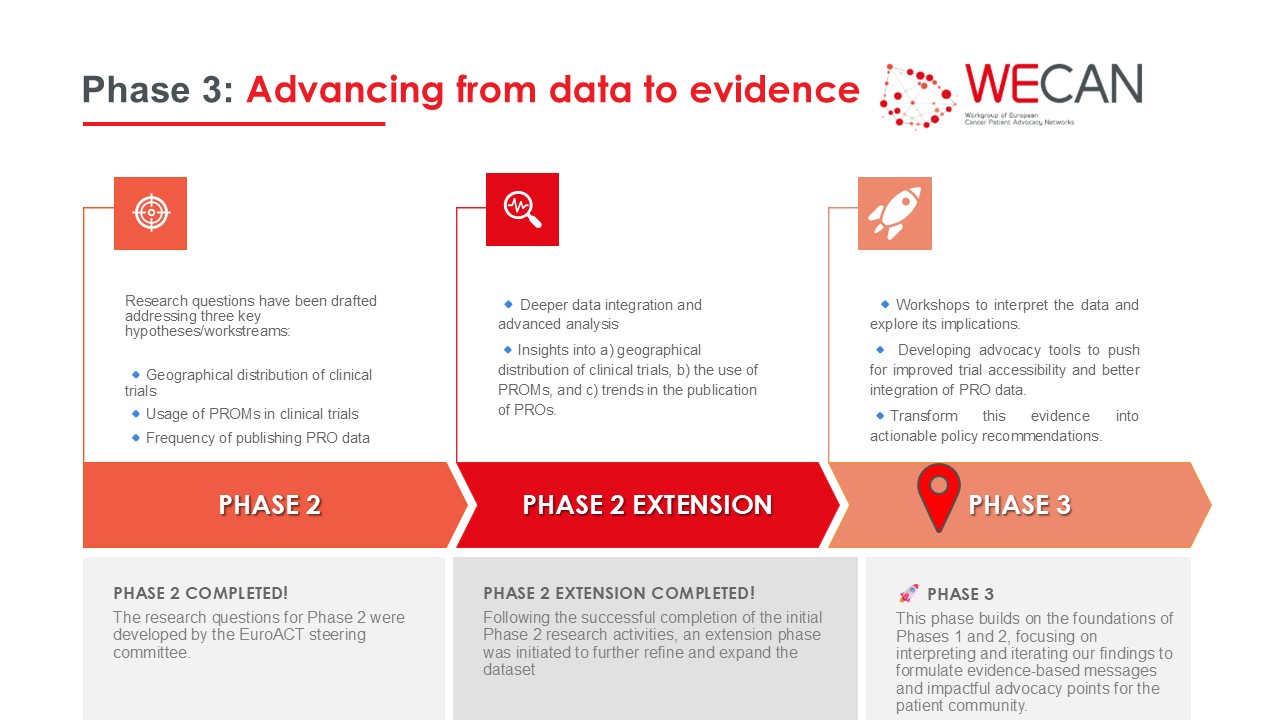
EuroACT Phase 3: Conversion – Data becomes evidence is well underway! This phase builds on the foundations of Phases 1 and 2, focusing on interpreting and iterating our findings to formulate evidence-based messages and impactful advocacy points for the patient community.
Key elements of Phase 3
1. Results interpretation
- • A critical assessment of results was conducted, synthesising research findings for each hypothesis and including a gap analysis exploring both identified gaps and their underlying causes, by our research partner Syreon.
- The EuroACT Steering Committee challenge meeting brought members together to prioritize findings, select actionable gaps, and develop aligned advocacy points.
2. Results iteration
- Focus group interviews workshops: We are conducting focus group interviews with key stakeholder groups, including:
- Patients and patient representatives/advocates
- HTA and PRO experts/developers
- Pharma organisation representatives
- Regulatory experts
These interviews will complement the “hard facts” from our data analysis with “soft facts” about the underlying motivations of decision-makers.
- A multi-stakeholder consensus interview workshop will bring diverse perspectives together to achieve alignment on barriers and actionable recommendations.
3. Results dissemination
Phase 3 will culminate in high-impact outputs:
- Publications: Scientific manuscripts, white papers, and conference presentations will share findings broadly.
This phase represents a crucial step in translating EuroACT data into concrete, evidence-based messages for the patient community and advocacy.
ABOUT THE PROJECT
EuroACT is a research project initiated by WECAN and the European Hematology Community. The project aims to understand the clinical trial landscape in the European region, based on data extracted from all relevant European clinical trial registers. Data from the past five years will unveil differences where clinical trials have been run in European countries and will describe how and which patient-reported outcomes (PROs) and quality-of-life (QoL) instruments have been used in clinical trials.


